Four medicines made by a Haryana-based firm in India have caused the death of 66 children in The Gambia due to the contaminants found in them. The World Health Organisation (WHO) has also issued an alert, stating that the cough syrups made by Maiden Pharmaceuticals Limited had diethylene glycol and ethylene glycol, which are toxic to human beings.
According to the details, medical officers in The Gambia raised alarm in July after several children began falling ill with kidney problems three to five days after taking a locally sold paracetamol syrup. By August, 28 had died, but health authorities said the toll would likely rise. Now 66 are dead, as per WHO.
Lab analysis confirmed “unacceptable” amounts of diethylene glycol and ethylene glycol in the Indian cough syrups, which can be toxic when consumed.
Read more: India’s aggressive regime is threatening South Asia’s peace: Pakistan
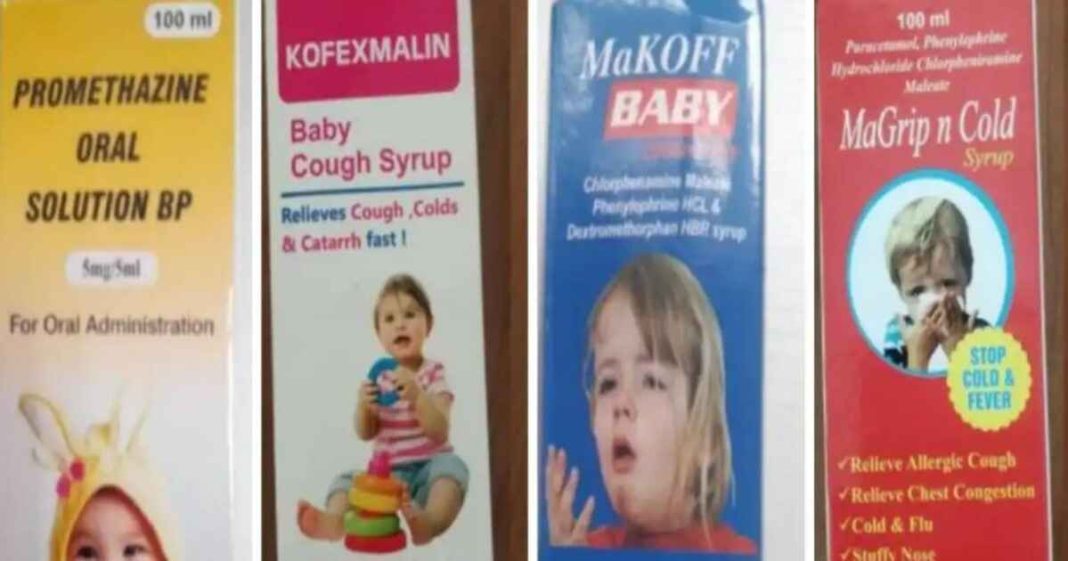
The four products are Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup, and Magrip N Cold Syrup, manufactured by Maiden Pharmaceuticals in India and later exported to countries in Asia, Africa, and Latin America, according to its website.
#WHO identifies 4 #paediatric cough syrups made by #Indian company Maiden Pharma as substandard
Alert: The products include Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup & Magrip N Cold Syrup #CoughSyrup #Gambia #MaidenPharmaceuticals pic.twitter.com/ToBgTJNG1n
— CNBC-TV18 (@CNBCTV18Live) October 6, 2022
“The substandard products referenced in this alert are unsafe and their use, especially in children, may result in serious injury or death,” said WHO in a statement.
Those substances are toxic to humans and can be fatal, it said, adding that the toxic effect “can include abdominal pain, vomiting, diarrhea, inability to pass urine, headache, altered mental state and acute kidney injury which may lead to death”.
WHO has also advised all the countries to remove the Indian cough syrups from their markets. WHO has pitched for increased surveillance within the supply chains of countries and regions likely to be affected by these products.
Read more: The suffering of masses by fake medicines
Interestingly, there has been no word or advice from India’s Central Drugs Standard Control Organisation (CDSCO) or its head, the Drugs Controller General of India (DCGI), nor has any statement been issued by India’s health ministry.