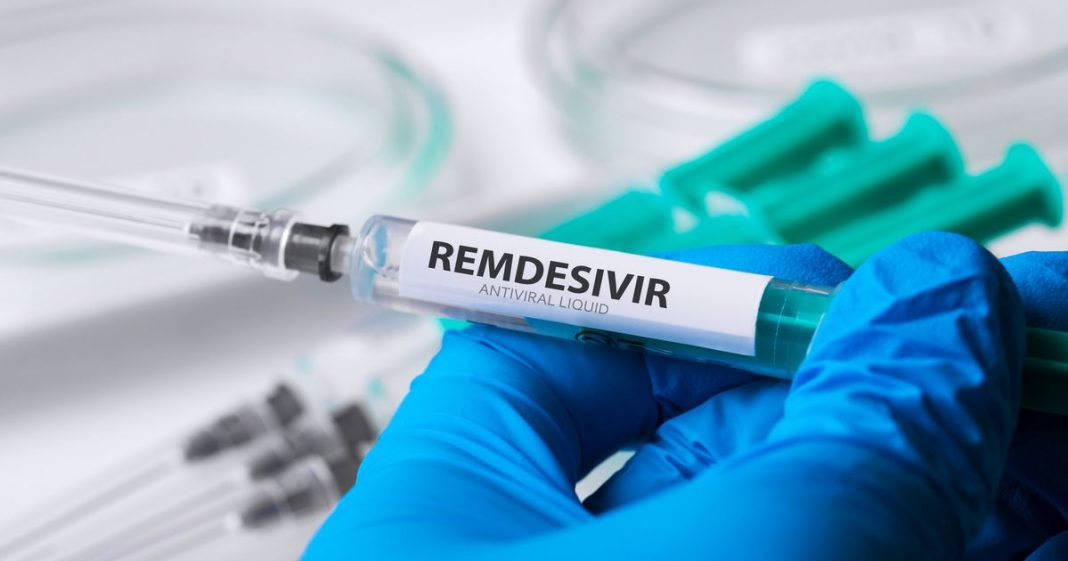
Anti-viral drug Remdesivir cuts recovery times in coronavirus patients, according to the full results of a trial published Friday night, three weeks after America’s top infectious diseases expert said the study showed the medication has “clear-cut” benefits.
Complete results from the research, which was carried out by US government agency the National Institute of Allergy and Infectious Diseases (NIAID), were published by leading medical periodical the New England Journal of Medicine.
In this double-blind RCT in 1063 adults hospitalized with #COVID19, a 10-day course of intravenous remidesivir was superior to placebo in shortening the time to recovery. The estimates of mortality at 14 days were 7.1% in the remdesivir group and 11.9% in the placebo group.
— NEJM (@NEJM) May 22, 2020
Remdisivir: Nations rapidly allowing its use in emergencies
The United States authorized the emergency use of remdesivir in hospitals on May 1, followed by Japan, while Europe is considering following suit.
Read more: US gives green light for ‘Remdesivir’ to be used as anti-corona drug
The study found that remdesivir, injected intravenously daily for 10 days, accelerated the recovery of hospitalized COVID-19 patients compared to a placebo in clinical tests on just over a thousand patients across 10 countries.
Encouraging results for Remdesivir
On April 29, NIAID director Anthony Fauci, who has become the US government’s trusted face on the coronavirus pandemic, said preliminary evidence indicated remdesivir had a “clear-cut, significant and positive effect in diminishing the time to recovery.”
The National Institutes of Health, of which the NIAID is a part, said Friday in a statement online that investigators found “remdesivir was most beneficial for hospitalized patients with severe disease who required supplemental oxygen.”
Remdesivir is not godsend, caution researchers
But the authors of the trial wrote that the drug did not prevent all deaths.
“Given high mortality despite the use of remdesivir, it is clear that treatment with an antiviral drug alone is not likely to be sufficient,” they added. “Future strategies should evaluate antiviral agents in combination with other therapeutic approaches or combinations of antiviral agents to continue to improve patient outcomes in Covid-19.”
Read more: Remdesivir alone is not enough, researchers conclude in first major Covid-19 trial of the drug
About 7.1 percent of patients given remdesivir in the trial group died within 14 days — compared with 11.9 percent in the placebo group.
However, the result is just below the statistical reliability threshold, meaning it could be down to chance rather than the capability of the drug.
Coronavirus: The situation so far
As of today, the global number of people infected by the Novel Coronavirus and suffering from the associated diseases COVID-19 has crossed 4.97 million. There have been 327,000 deaths associated with the disease. A statistic to take heart from is the fact that 1.89 million people suffering from COVID-19 have recovered.
COVID-19 associated lockdowns have caused a slowdown in the international economy, with experts saying that it will shrink by as much as 6% this year. Estimates of its recovery do not show it recovering before 2022.
AFP with additional input by GVS News Desk