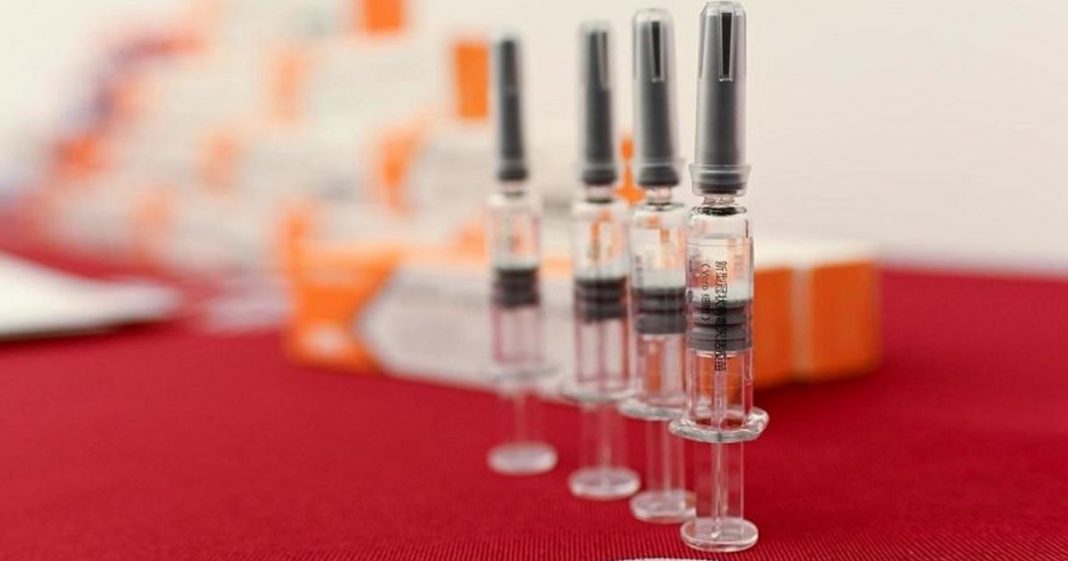
India has authorized the emergency use of two coronavirus vaccines developed by AstraZeneca and Oxford University and by local pharmaceutical firm Bharat Biotech, the country’s drug regulator said Sunday.
“The… vaccines of Serum Institute (AstraZeneca/Oxford vaccine) and Bharat Biotech are being approved for restricted use in emergency situations,” the Drugs Controller General of India, V.G. Somani, said at a briefing.
Prime Minister Narendra Modi tweeted that the fast-track approvals were “a decisive turning point to strengthen a spirited fight” that “accelerates the road to a healthier and Covid-free nation”.
It would make every Indian proud that the two vaccines that have been given emergency use approval are made in India! This shows the eagerness of our scientific community to fulfil the dream of an Aatmanirbhar Bharat, at the root of which is care and compassion.
— Narendra Modi (@narendramodi) January 3, 2021
Read more: India prepares for mass COVID-19 vaccine drive
India is the world’s second most-infected nation with more than 10.3 million cases and almost 150,000 deaths, although its rate of infection has come down significantly from a mid-September peak of more than 90,000 cases daily.
The approval is expected to kick off one of the world’s biggest vaccination drives in the country of 1.3 billion people.
The government has already been holding nationwide drills ahead of the mass inoculation drive and 96,000 health workers have been trained to administer the shots.
Read more: India’s coronavirus cases pass 10 million
A government panel on Friday recommended emergency use of the AstraZeneca-Oxford University shot and the first injections could be given in the coming week after the Drugs Control Authority of India gives final approval.
We reiterate our gratitude to doctors, medical staff, scientists, police personnel, sanitation workers and all Corona warriors for the outstanding work done, that too in adverse circumstances. We will remain eternally grateful to them for saving many lives.
— Narendra Modi (@narendramodi) January 3, 2021
Somani added to reporters after the briefing that the drug regulator would “never approve anything if there is the slightest safety concern”.
“The vaccines are 100 percent safe,” he said, adding that side effects such as “mild fever, pain, and allergy are common for every vaccine”.
Read more: Pakistan, India suspend flights from UK amid new COVID-19 strain
Serum Institute of India, the world’s biggest vaccine producer, has already stockpiled tens of millions of doses of AstraZeneca’s Covishield ready for the campaign and 96,000 health workers have been trained for the inoculation drive.
Serum Institutes chief executive Adar Poonawalla tweeted after the approvals that the vaccine would be “ready to roll-out in the coming weeks”.
Britain and Argentina this week authorized the AstraZeneca vaccine while the World Health Organization on Thursday granted emergency validation to the rival Pfizer-BioNTech vaccine.
Read more: India in no position to talk about minority rights violations anywhere: Pakistan